Imaging entire intact human organs to understand the spatial relationships and 3D morphology at a cellular scale is one of key goals of biomedical imaging. In recent years, several techniques have been used to image intact organs at various length scales; however, they are not without limitations. Many techniques that provide single-cell resolution involve mechanical slicing of tissues into thin sections that distort the tissue and also hinder accurately reconstructing samples with larger volumes. Tissues inside our body are also naturally opaque, which limits its visualization. This has been tackled using advancements in tissue clearing which refers to chemical techniques to make the tissues transparent, which can then be subjected to high resolution 3D imaging methods, including light-sheet microscopy. While optical clearing of whole adult human organs has been recently achieved, it often requires several months which leads to changes in morphology. Other techniques include magnetic resonance imaging (MRI) that requires minimal tissue preparation and causes minimal tissue distortion. But this type of scan can require up to 100h of scanning for the entire human brain and does not provide cellular resolution. Multi-beam electron microscopy is another technique that can provide tissue images at a cellular scale; however, currently it cannot be employed on large volumes of tissues of an intact organ.
The large size of adult human organs also presents a fundamental imaging challenge: the light needs to penetrate through the depth of the tissue during imaging and diffusion of the reagents is also limited due to the thickness of an organ.
Figure 1: (A) Intact human brain mounted in a polyethylene terephthalate jar with ethanol–agar stabilization and with the reference jar on top. (B) Maximum intensity projection of a whole human lung with two randomly selected volumes of interest. (Images from Walsh et al, 2021, reused under CC license)
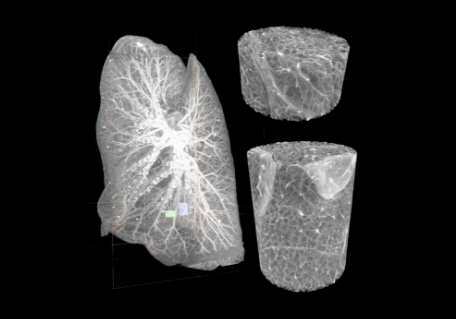
In a recent study published in Nature, researchers at University College London employed a method which they termed Hierarchical phase-contrast tomography (HiP-CT) using a high-energy, fourth-generation, a synchrotron that is the source of world’s brightest X-rays which can achieve a 100-fold increase in brilliance compared to the previous versions. Using this method, the researchers could image five intact human organs: brain, lung, heart, kidney, and spleen.
This method requires specific sample preparation where the organs are fixed, partially dehydrated, and physically stabilized for scanning. Due to the mild nature of sample preparation and its non-destructive imaging, the samples could also be compared with other imaging techniques. This method was also used to perform 3D imaging of a SARS-CoV-2 infected lung. Quantitative analysis of the lung architecture aligned with the pathological signature where there is an increased volume of ventilated air in the lungs that does not participate in gas exchange.
Recently, several international efforts have been initiated to map every cell in the human body, including the Human Cell Atlas, and Human Biomolecular Atlas Program. Such advances in imaging technologies that can map spatial organization and morphology of individual cells up to the scale of intact organs can contribute tremendously to these efforts. This in turn can provide unprecedented insights to systems-level behavior in health or disease.